HBCU presidents encourage Black people to participate in COVID-19 vaccine trial
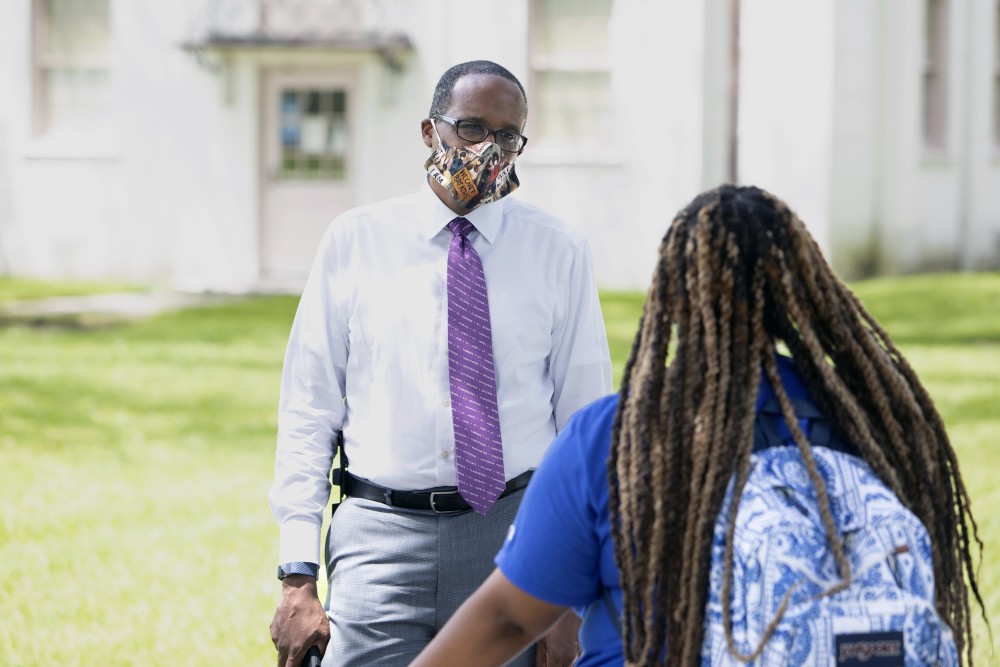
On a recent Sunday, photos of two Black men graced the sanctuary of First Grace United Methodist Church in New Orleans as Shawn Moses Anglim spoke during “a little children’s moment.”
The two church members pictured, Dillard University president Walter Kimbrough and pediatrician Ted Atkinson, were lifted up as role models for the children because they are participating in a COVID-19 vaccine study at Ochsner Medical Center in New Orleans.
“Everybody in the world is suddenly connected,” Anglim said in an interview. “We all need this vaccination, and we need our medical community to develop it.”
However, distrust of the medical community has resulted in a shortage of African Americans willing to be research subjects. This could undermine the effectiveness of any vaccine developed to fight the coronavirus.
In a joint open letter to their campus communities, Kimbrough and Xavier University of Louisiana president C. Reynold Verret appealed to students, faculty, and staff to consider participating in the study.
“The people and communities we serve look to us as an example,” the two leaders of historically Black universities said in the September 2 letter.
Kimbrough said Verret recruited him to participate in the study.
“He said we could encourage people,” Kimbrough said. “Some scientists are concerned that any vaccine that’s developed might not be effective on African Americans, who are disproportionately impacted, given that they haven’t been tested on them.”
Read our latest issue or browse back issues.
Some African Americans don’t trust medical researchers, and there are understandable reasons for that, he said.
“There are plenty of historical examples of mistreatment, ethically problematic cases of treatment bias and mistreatment of African Americans,” Kimbrough said.
The first thought some Black people have in response to requests to participate in a study is the Tuskegee Syphilis Study, said Claude Tellis, a retired doctor and member of Camphor Memorial United Methodist Church in Baton Rouge, Louisiana. From 1932 to 1972, the effects of syphilis were studied in Black men at Tuskegee Institute in Alabama. The men were told they were being treated, which wasn’t true.
Tellis, who cochaired a forum with Ochsner Medical Center to encourage people in the Black community to participate in the vaccine trial, said there are other examples, too.
J. Marion Sims, a 19th-century doctor known as the father of gynecology, performed surgical experiments on enslaved black women without using anesthesia. A statue honoring Sims in New York City’s Central Park was removed in 2018.
Cancer cells from Henrietta Lacks, a Black woman who died in 1951 of cervical cancer, were used for research without her consent. Neither she nor her family received any compensation.
And as recently as this September, the Department of Homeland Security announced an investigation into a whistleblower’s claim that federal immigration detainees in Georgia were given gynecological surgeries, including hysterectomies, without their informed consent.
“That just makes everything worse,” Tellis said. “That would be the kind of thing that would keep people from participating in studies.”
Furthermore, a 2001 study found that Black people get subpar medical treatment in general and have worse outcomes than White people.
“Even after controlling for access to care, insurance, and location, minorities got lower quality care in the health-care system,” said Derek Griffith, director of the Center for Research on Men’s Health at Vanderbilt University. “That’s been a consistent fact that’s been known and documented for at least 20 years.”
In their letter urging participation in COVID-19 studies, Kimbrough and Verret pointed out that “today there are many regulations in place to assure the ethical execution of medical studies, including oversight by human subjects’ committees with diverse membership and participation of clinicians of color.”
Kimbrough said his first meeting as a study participant was three hours of orientation explaining how the study works.
“You spend an hour and a half going through tests and reviewing a video and all that,” Kimbrough said. “They keep saying that any time you want to stop participating, you can.”
Still, mistrust about participating in medical studies continues among African Americans, Kimbrough said.
“It’s a problem, and the medical community has got to continue to work with historically Black colleges and universities, Black churches, Black civil rights groups, and Black political leaders to really have more of these conversations.” —United Methodist News Service






